-
About Stone

-
-
-
-
NEWS
NEWS
Guangdong Stone significant changes approved by CDE
2023-06-14
The oral liquid medicinal PET bottle (registration no., B20190008708), external liquid medicinal PET bottle (registration no., B20190007921), external liquid medicinal HDPE bottle (registration no., B20190007907) and liquid medicinal PP spray pump (registration no., B20190006141), the major changes in name and production address of these four series were approved by the Center for Drug Evaluation (CDE) of the State Drug Administration in June 2022. Including the oral liquid pharmaceutical HDPE bottle (registration no., B20190008709) approved in December 2021, the owners of these five series with registration status A have all been transferred to Guangdong Stone Medicinal Packaging Materials Co., Ltd. Guangdong Stone will officially provide products and services to domestic pharmaceutical companies after it owns these five series with the registration status of A. This is a milestone for the 16-year-old Stone. The capacity will be improved highly to realize the transformation of small and medium-sized enterprises into large enterprises.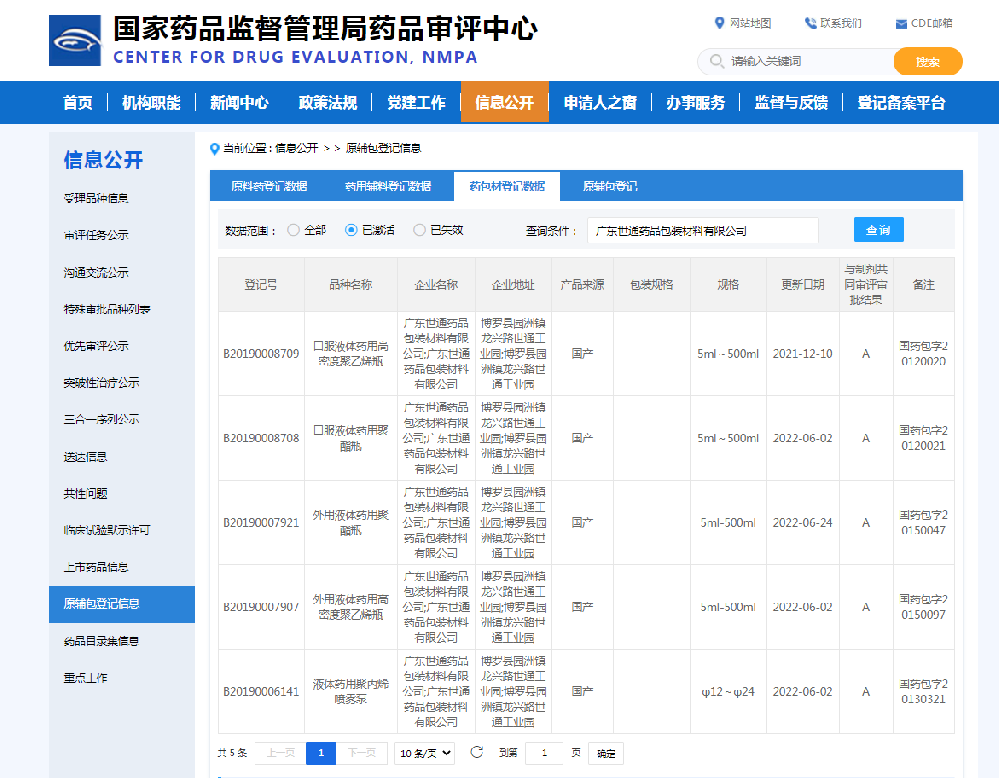
热门文章
2023-06-14

广东世通药品包装材料有限公司
Guangdong stone medicinal Packaging Material Co., Ltd

JOIN US
Stone Industrial Zone, Longxing Road, Yuanzhou Town, Boluo County, Huizhou City, Guangdong Province, 516123 China
0752-666 2682



















